- Adverse events for those taking Mallinckrodt Pharmaceuticals‘ drug Acthar spiked last year, according to FDA data. Hospitalizations tripled, and “other serious” side effects quadrupled. In a lawsuit, a former Mallinckrodt sales rep says he was pushed to promote Acthar for off-label uses. A neurologist who has studied the drug says the spike in hospitalizations could be a sign it is being overused.
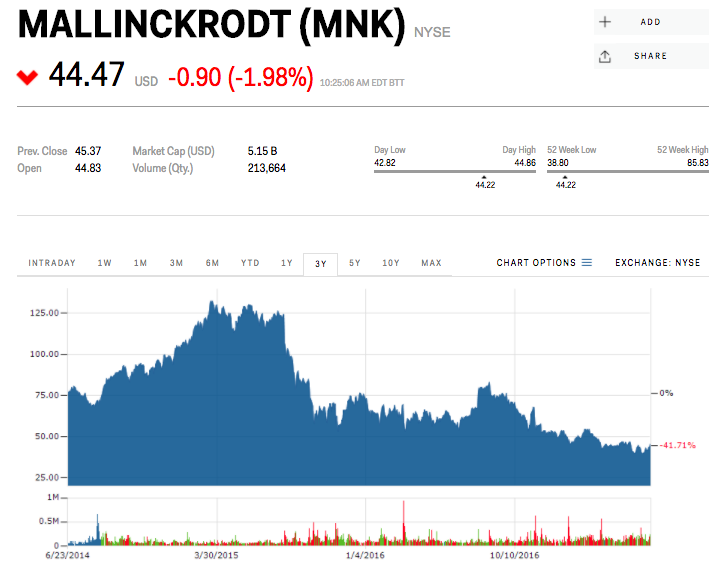
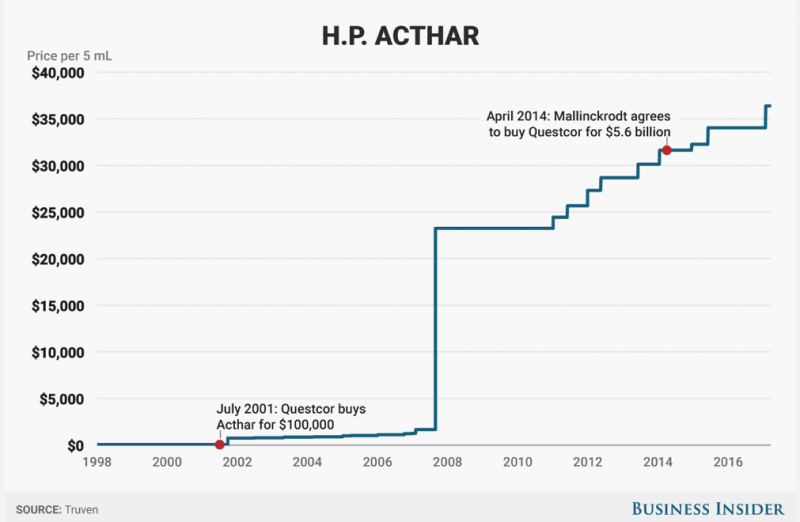
Mallinckrodt Pharmaceuticals and its $36,382 blockbuster drug – a decades-old treatment called Acthar – are under siege from short sellers.
These traders – who are profiting from the stock’s decline – have questioned the drug’s efficacy, Mallinckrodt’s dependence on deals with pharmaceutical middlemen for the drug’s expanding use, and why a drug that is primarily indicated as a treatment for infants is turning into a massive expense for Medicare, a program for the elderly.
Now the critics have a new line of attack. Data from the Food and Drug Administration shows that reports of adverse events for those taking the drug – everything from hospitalization to death – have spiked recently. The increase is far faster than the rate of prescriptions.
Andrew Left of Citron Research pounced on the data, releasing a report Thursday saying Citron “believes the reason for the increase in adverse events is the company’s expansion of Acthar into new indications where it has not conducted clinical trials in order to test its safety in new indications.”
He's not the only short seller targeting the company, but he is the shrillest. Mallinckrodt shares tumbled in early trading Thursday before regaining their losses.
To be clear: The raw numbers are relatively small, with only about 9,000 people taking Acthar, according to Mallinckrodt.
Eighty-two deaths were reported in 2016, up from 69 the year before. Hospitalizations nearly doubled to 427, and "other serious" side effects quadrupled to 436.
The company released a statement saying that side effects linked to the drug were often relatively minor and that correlation of side effects with Acthar didn't necessarily mean causation. You can read that here. 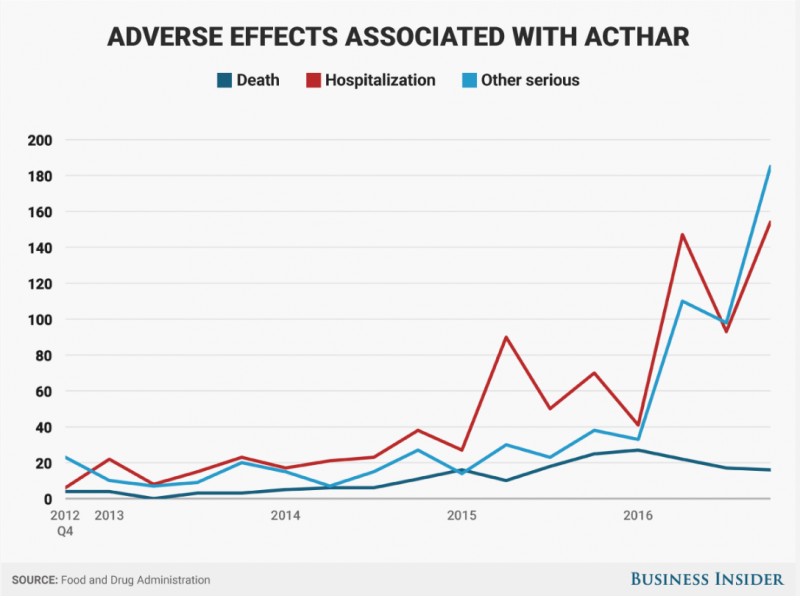
What the data does show us is that Mallinckrodt's strategy of taking an old drug that never went through the rigors of FDA approval and trying to expand its use could be running into serious limits. The company has turned Acthar into a drug with over $1 billion in annual sales by persuading doctors to prescribe it even to the sickest patients and even when there are much cheaper alternatives.
I asked Dr. Dennis Bourdette, the chair of neurology at the Oregon Health & Science University, about the FDA data. He has studied Acthar's use - and he's not a fan.
To Bourdette, the spike in adverse events could be a sign the drug is being overused in a way that is triggering side effects.
"Some doctors are using Acthar like an ongoing, monthly, or weekly therapy over long periods of time, and when you do that there are all sorts of side effects that can occur because of the steroid effects that can occur," Bourdette said.
On Acthar's website, the company takes pains to say it is not a steroid, even though it "works by helping your body produce its own natural steroid hormones." Bourdette says that doesn't matter because it still acts like one. "ACTH is having all these effects on the immune system apart from releasing cortisol," Bourdette said, referring to the adrenocorticotropic hormone in Acthar.
"When your body produces too much cortisol you're going to have chronic steroid side effects we've known about for decades, and every doctor is trained in those side effects."
'No idea'
So why are the doctors prescribing so much Acthar?
"I have no idea," Bourdette said.
Acthar is supposed to be taken for about five to 15 days to treat infantile spasms and multiple sclerosis, but there's no specification on dosage for the 17 other conditions that Mallinckrodt says can be treated with Acthar.
The drug was grandfathered into FDA approval because it dates back to the 1950s before clinical testing was required. As such, some have questioned its efficacy.
To be fair, Mallinckrodt has said repeatedly that only the most desperate patients take Acthar, suggesting it's not the primary treatment for many of the ailments it is used against. Instead, the company says, it is used only after other drugs don't work.
Mallinckrodt also says on its website that while "the exact way that Acthar works in the body is unknown, further studies are being conducted" and that some information on the website "is based on laboratory data, and how it relates to patient benefit is unknown."

Some ideas
So why would doctors prescribe too much Acthar for the wrong ailments? We can't be sure, but we do know that in December a former Acthar sales rep, Barry Franks, filed a whistle-blower suit against Mallinckrodt.
Franks accused the company of pushing him to promote Acthar's use for off-label indications - in fact, he's saying his bonus depended on it. He says when he wouldn't do things Mallinckrodt's way - and his sales team didn't see the "explosive growth" other teams did - he was fired.
"Franks ... alleges that there were illegal sales practices being committed in other regions, and that Mallinckrodt used these regions as the gold standard or basis Incentive Bonus Plans for which all regions were compared and asked to duplicate. Franks was expected to deliver sales that were not supported by lawful practices.
...
"Franks was also aware of other compliance related issues at Mallinckrodt, which Franks ... alleges that these issues include but were not limited to: potential insurance/Medicare fraud ... HIPPA violations where four or eight week prescriptions were provided where there was no patient visit and violations where Mallinckrodt permitted, for a certain period of time, certain employees to manipulate the compensation plan by having physicians wrote shorter prescriptions that were refilled, to earn a bonus on the patient at the shortest prescription interval.
"(Shorter prescriptions did not allow sufficient time to see if a patient responded to the drug. In some cases the drug was shipped the same day as the referral was received. This was done to 'game the system' and potentially commit insurance/Medicare fraud.)"
Mallinckrodt didn't respond to a request for comment on Franks' claims.
Another reason doctors may want to prescribe Acthar goes back to Bourdette's study on the few doctors who do prescribe Acthar. Mallinckrodt, like a lot of other drug companies, also has a speakers program through which it can book and pay doctors to talk about its drugs.
Typically, the top prescribers of Acthar not only have received payments from Mallinckrodt but also "are typically getting larger payments from companies that are developing disease-modifying therapies," Bourdette told us back in March.
One top prescriber, Dr. Regina Berkovich, an assistant professor of clinical neurology at the University of Southern California, accepted $23,895 for writing a paper and talking about Acthar in 2013. That was out of $142,978 in speaking and other fees from various drug companies she collected that year.
Another doctor, Robert Baughman, a professor of internal medicine at the University of Cincinnati, is collaborating with Mallinckrodt on a study on Acthar's efficacy curing patients with sarcoidosis, a disease that affects the lungs and lymph glands. This is part of the "investigator-initiated research," which Mallinckrodt supports financially through grants.He has also received $21,625 for promoting Acthar from 2013 to 2015 (that's out of $116,000 for speaking and promotion fees for drugs during that period). He declined to comment on his research when we asked him about it March.
What he's published about Acthar is not encouraging. Of 47 patients, 18 discontinued the program because of "cost (four patients), death (two patients), or drug toxicity (eleven patients), or noncompliance (1 patient)."
Still, both his study and Berkovich's study are being touted as advancements by Mallinckrodt.
The No. 1 prescriber of Acthar (making up 1% of all prescriptions) is an Ohio rheumatologist named David Mandel, according to the most recent government data. In August of 2014, he was forced to pay a $640,000 fine for the "shipment of misbranded drugs." From 2013 to 2015, Mandel has received $22,762 in speaking and other fees from Mallinckrodt for promoting Acthar.
Until rigorous, independent clinical trials are done, there will continue to be questions about the efficacy of Acthar. At least, there should be for the sake of anyone taking it.